
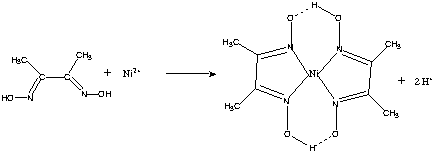
Except where otherwise noted, data are given for materials in their (at 25 °C 77 °F, 100 kPa).Y ( Y N?)Dimethylglyoxime is a described by the formula CH 3C(NOH)C(NOH)CH 3. Its abbreviation is dmgH 2 for neutral form, and dmgH for anionic form, where H stands for hydrogen. This colourless solid is the di derivative of the diketone butane-2,3-dione (also known as ). DmgH 2 is used in the analysis of. Its are of theoretical interest as models for enzymes and as catalysts. Many related ligands can be prepared from other diketones, e.g.Preparation Dimethylglyoxime can be prepared from first by reaction with to give. The second is installed using sodium monosulfonate:Complexes Dimethylglyoxime is used to detect and quantify, which forms the bright red complex (Ni(dmgH) 2).
Hello, I would really appreciate some help. I had to do a gravimetric analysis of Nickel in Ore. My question is, how can I determine the percent by mass of the Nickel having only the mass of the Nickel complex (Ni(DMGH2) and the ore mass? I don't know how to find the mass of only the Nickel from the Nickel complex. If anyone knows, please help!!! CID 83518 C8H14N4NiO4 CID 83518 - structure, chemical names, physical and chemical properties, classification, patents, literature, biological activities, safety.
The reaction was discovered by in 1905.Cobalt complexes have also received much attention. In the macrocycle dmgH 2 2− mimics the macrocyclic ligand found in.
Metal-organic frameworks (MOFs) have been tremendously used as photocatalysts for H 2 generation in recent years. Lacking native active sites (so-called co-catalyst) for H 2 generation motivates the incorporation of noble metals and their molecular complexes, hydrogenase active site mimics into MOFs to promote H 2 generation.
We herein report an noble-metal-free photocatalytic H 2 generation system consisting of Erythrosin B dye-sensitized MIL-101(Cr) as a light absorber and Ni(dmgH) 2 as a co-catalyst. It is found that Ni(dmgH) 2 can serve as an efficient co-catalyst to boost H 2 generation in the presence of triethanolamine (TEOA) as an electron donor under visible light irradiation. The optimal MIL-101(Cr)/Ni(dmgH) 2 hybrid (5 wt% Ni(dmgH) 2) displays a hydrogen H 2 rate of 45.5 μmol h −1, which is 10 times greater than the control sample without Ni(dmgH) 2 loading. This paper provides a novel design route for active H 2 generation systems by combining molecular complexes of earth-abundant metal and MOFs photocatalysts.
Yupeng Yuan is an associate professor in school of Chemistry & Chemical Engineering, Anhui University, China. He received his BE (2002) and ME degrees (2005) from Hefei University of Technology and PhD degree (2008) from Nanjing University. He worked as a Research Fellow at Nanyang Technological University in 2012–2013 and researcher scholar at Georgia Institute of Technology in 2016. His current researches focus on photocatalytic water splitting, photocatalytic CO 2 conversion to hydrocarbon fuels, photoelectrochemistry and first principle calculations.
